The definition of hybridization according to experts, among others, are as follows Understanding Hybridization According to Experts When two atoms will be chemically bonded, the two atoms need an empty orbital to be occupied by electrons from each of these atoms so that after binding, both atoms will occupy the same orbitals on their valence electrons. Therefore, in the hybridization process involves the configuration of electrons, especially the valence electrons used for binding.
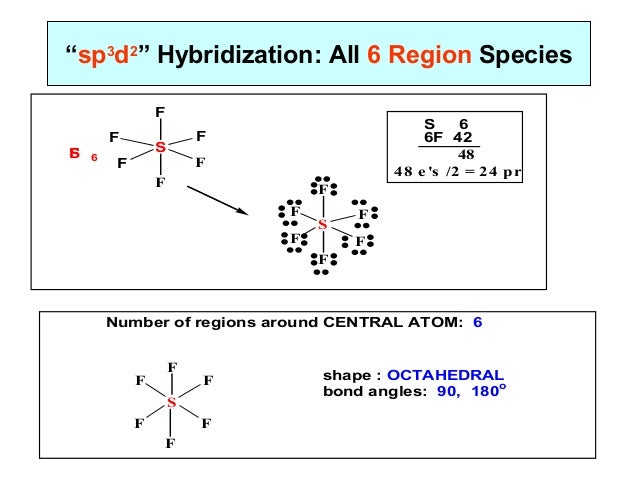
Sp3d angle series#
Hybridization can be interpreted as a series of processes combining orbitals from one atom with another atom when the meaning of a chemical bond occurs so as to achieve lower energy or high stability.
The hybridization theory explains the bonding to alkenes and methane. The number of p characters, which is decided mainly by hybridization of orbitals, can be used to predict molecular properties such as acidity or basicity. The hybridization theory is an integral part of the meaning of organic chemistry , one of the most interesting examples is the Baldwin rule. To draw the reaction mechanism it is sometimes necessary to draw a classic bond with two atoms sharing two electrons. This concept was developed for simple chemical systems, but this approach was later applied more broadly, and today is considered an effective heuristic for rationalizing the structure of organic compounds. It provides a simple orbital image that is equivalent to Lewis’s structure. In fact, methane has four bonds of equal strength separated by a tetrahedral bond angle of 109.5 °. Pauling explains this by supposing that in the presence of four hydrogen atoms, s and p orbitals form four equivalent combinations or hybrid orbitals, each symbolized by sp3 to show its composition, which is directed along the four CH bonds.
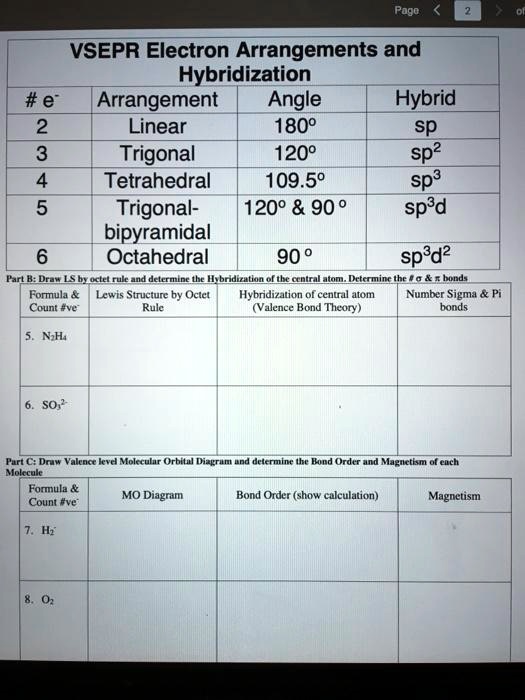
Sp3d angle free#
So it can be “concluded” that the carbon atom will form three bonds at right angles (using p orbitals) and the fourth weaker bond using s orbitals in the free direction. The chemist Linus Pauling first developed the theory of hybridization in 1931 to explain the structure of simple molecules such as methane (CH4) using atomic orbitals. Pauling shows that carbon atoms form four bonds using one and three p orbitals. Understanding Hybridization According to Experts.

those in the 2s and 2p sublevels are available to form chemical bonds with other atoms. 2s orbitals can hold up to two electrons, and there are three 2p orbitals, each capable of holding up to two electrons, which means that 2p orbitals can hold up to six electrons.

Hybridization occurs when orbitals are in atomic theory mix to form new atomic orbitals. New orbitals can hold the same total number of electrons as the old ones. The properties and energy of the new hybridized orbitals are ‘averages’ from the original uncarbonized orbitals. The concept of hybridization was introduced because that is the best explanation for the fact that all C-H bonds in molecules such as methane are identical.įor example, in its basic state, carbon atoms naturally have an electron configuration of 1s 2 2s 2 2p 2 . Four outer electrons, i.e.
